39Ar/40Ar experiments are sometimes used to derive Ar diffusion parameters from 39Ar release in vacuo assuming Fickian diffusion. This diffusivity is then used to interpret ages as Tt points which are then used to model tectonic evolution. While in K-feldspars from plutonic rocks this has recently shown to be an invalid approach (Villa, 1994) the currently unsolved question is if any K-feldspar at all follows Fick's Law. If Ar diffusion only depends on temperature, Fick's Law might be a viable model, but if the other parameters (extended defects, multipath diffusion, atomistic effects) also play a role, then less simplified interpretive models must be sought. Three small (×1-3 mg) chips from a cm-size gem-quality orthoclase crystal from Madagascar were subjected to different stepwise heating experiments:
(a) irradiation with 7x1017 n/cm2 followed by conventional stepwise heating, with a monotonic temperature increase from 600° to 1650°C
(b) irradiation followed by isothermal degassing at 750°C in 10 steps over 20 hours, followed by steps at increasing temperature up to 1650°C
(c) conventional stepwise heating, with a monotonic temperature increase from 600° to 1650°C, without previous irradiation.
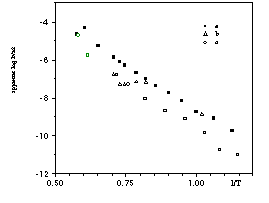
From a and b it is possible to obtain apparent diffusivities both for 39Ar and 40Ar* (radiogenic 40Ar). Since the step ages of the Madagascar orthoclase are practically constant, the two apparent diffusivities agree with each other within the size of the symbols in Fig.1. Fig. 1 therefore only shows the 40Ar* diffusivities for a, b and c.
The activation energy of a is 172 kJ/mole, defined by a well-correlated Arrhenius line in Fig. 1. A significant portion of the gas (>70%) is released above the nominal melting temperature of orthoclase, implying that the "liquid" is very viscous and melting probably starts by far-from-complete depolymerization of the solid crystal.
The behaviour of b is quite different. Diffusivities in the 10 isothermal steps first drop (the same non-Fickian behaviour shown by Villa (1994) by 0.65 log units, then gradually rise again by 1 log unit. The diffusivities of the steps at increasing temperature are considerably lower than those of a and decrease with increasing T. Structural modification is an attractive explanation for the delayed Ar release. The steps above 1050°C lie on a line with an unreasonable activation energy of 547 kJ/mole; this may suggest that the high activation energy reported for cycled heating (Arnaud and Kelley, 1996) also may have been biased by the structural modification. The 20 hour-heating at 750°C is clearly seen also to affect the 39Ar release above 1200°C and to delay Ar release to 1500°C; the residual structure in the molten feldspar appears not to have recovered the structural modifications of the prolonged 750°C heating.
The unirradiated feldspar c shows a linear behaviour like a; however, the activation energy, 189 kJ/mole, is significantly different. The diffusivities are lower by × 1 order of magnitude over the 600°-1000°C range.
If the release of Ar from the Madagascar orthoclase is interpreted in terms of "traps" (Arnaud and Kelley, 1996), it is necessary to envisage two entirely different populations of traps: one present over geologic times, in which excess Ar is located, and one produced by the irradiation, which accounts for the 10% decrease in the activation energy. Another possibility is interpreting the excessively high 40Ar*/39Ar ratios in the first steps (ascribed by Arnaud and Kelley, 1996, to excess Ar) as due to the recoil of 39Ar (0.1 µm) accompanied by fast pathway diffusion of 40Ar* along the neutron damage tracks in the irradiated crystals.
In any case, there are extremely strong indications that not even gem-quality K-feldspars follow Fick's Law of diffusion in laboratory experiments.
Arnaud, N.O. & Kelley, S.P., EOS, Trans. AGU. in press (abs.) (1996).
Villa, I.M., Earth Planet. Sci. Lett. 122, 393-401 (1994).

Fig. 1.