Clinopyroxene has higher mineral-melt partition coefficients for REE and HFSE than other common mantle phases and often controls their abundances in anhydrous mantle melts. Accurate partition coefficients for these elements are crucial for reliable modeling of partial melting processes, for linking primitive melts to potential sources represented by entrained xenoliths, and for distinguishing between mantle source regions in an effort to understand the scale and geometry of mantle heterogeneities (e.g., La/Nb ratio) (McKenzie and O'Nions, 1995). Several recent studies (Hauri et al., 1994; Forsythe et al., 1994; Skulski et al., 1994; Gaetani and Grove, 1995; Hack et al., 1994) have demonstrated the importance of melt structure and crystal chemical controls on partitioning behavior of REE and HFSE between clinopyroxene and basaltic melts, with HFSE incorporated into the M1 site and charge balanced with tetrahedral Al (Skulski et al., 1994) and REE3+ incorporated into the M2 site, replacing Ca2+ via coupled substitution with Na+(Gaetani and Grove, 1995). We investigated partitioning of Ti and Na between cpx and alkalic mafic liquids from 1 atm to 18 kbar to establish the pressure dependency of these relationships. The behavior of the minor element Ti is taken as analogous to the behavior of HFSE (Forsythe et al., 1994). Na is significant as an index of REE partitioning, and because it can function as either network former or modifier in silicate melts (i.e., Na/Al ratio) (Hess, 1980). In addition, McKenzie and O'Nions (1995) note that the partitioning of Na between melt and high-Ca cpx is a strong but poorly understood function of pressure .
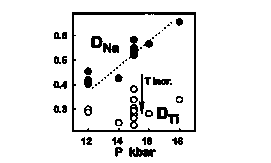
We performed experiments on an alkalic basalt (9.6 wt.% MgO) and nephelinite (8.8 wt. % MgO) from the Rungwe volcanic province, African Western Rift (Furman, 1995). One atmosphere experiments were conducted at the University of Virginia using a gas-mixing vertical tube furnace with fO2 controlled via CO2-H2 mixtures near QFM. Experiments at 12 to 18 kbar were done in a piston cylinder apparatus at MIT. Temperatures for moderate pressure experiments ranged from 1260 to 1410°C; clinopyroxene and melt were the only phases observed over this interval (maximum degree of crystallinity ~30 vol.%) for both starting compositions. At constant pressure we find a positive correlation between Ti and Na in cpx, both increasing with decreasing T. DNa is strongly P dependent while DTi is not significantly P dependent (see figure). Prediction of HFSE partitioning using parameterizations of D that take into account compositional variations only (Forsythe et al., 1994; Skulski et al., 1994; Gaetani and Grove, 1995; Hack et al., 1994) is supported by this study. However, because REE may be incorporated into cpx via the coupled substitution with Na, our results indicate that compositional variables alone are not adequate to describe changes in DREE with P. We apply these results to the geochemistry of mafic lavas from the African Western Rift and suggest that a plume source is not required for magmatism in this area.
Forsythe, L.M., Nielson, R.L. & Fisk, M.R., Chemical Geology 117, 107-125 (1994).
Furman, T., Contrib. Mineral. Petrol. 122, 97-115 (1995).
Gaetani, G.A. & Grove, T.L., Geochim. Cosmochim. Acta 59, 1951-1962 (1995).
Hack, P.J., Nielson, R.L. & Johnston, A.D., Chemical Geology 117, 89-105 (1994).
Hauri, E.H., Wagner, T.P. & Grove, T.L., Chemical Geology 117, 149-166 (1994).
Hess, P.C., In Physics of Magmatic Processes (Hargraves, R.B. ed) 3-48 (Princeton University Press, 1980).
McKenzie, D. & O'Nions, R.K., J. Petrol. 36, 133-159 (1995).
Skulski, T., Minarik, W. & Watson, E.B., Chemical Geology 117, 127-147 (1994). 
Fig. 1: Partitioning of Na (solid circles) and Ti (open circles) between cpx and glass in experiments on alkali basalt from 12 to 18 kbar. The arrow shows direction of T dependence on D. Y-axis is Dcpx/glass (mole fraction element in cpx divided by mole fraction element in glass).