Studies on organic sulphur compounds (OSC) in sediments and oils have indicated that they are formed via the incorporation of inorganic sulphur species into functionalized lipids during early diagenesis. Simulation experiments of this hydrosulphurization reaction have shown that for example phytol in H2S saturated water is capable of forming C20 isoprenoid thiophenes (Fukushima et al., 1992). In this study we investigated the abiotic incorporation of sulphur into algal biomass of the prymnesiophyte Phaeocystis sp..
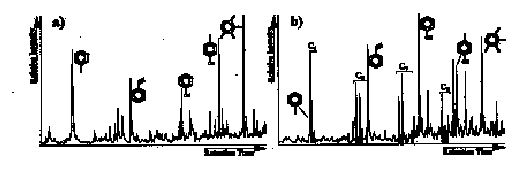
A simulation experiment of the natural hydrosulphurization reaction was performed using algal material of Phaeocystis sp. (collected during spring blooms in the Dutch Wadden Sea and North Sea in 1991 and 1992) as a substrate. The reaction was performed according to Schouten et al. (1993) for 4 weeks at 50ƒC. Fractions collected after extraction, saponification and acid hydrolysis - before and after sulphurization - were checked for the presence of OSC. Only low amounts of low molecular weight (LMW) OSC were detected, indicating that only very minor sulphur incorporation into lipids had occurred. The residues of the algal material were analyzed by flash pyrolysis-gas chromatography-mass spectrometry (Fig. 1). After 4 weeks of sulphurization thiophenes with LMW carbon skeletons are major pyrolysis products. As no LMW OSC were detected in the lipid fractions, this indicates that the OSC formed, must have been incorporated into the macromolecular matrix (MM). Therefore, the OSC must originate from compounds containing several functionalities necessary for sulphur incorporation and linking to the MM. Phaeocystis sp. can contain up to 90% of carbohydrate-rich mucus material during blooms (Rousseau et al., 1990). These carbohydrates may form an excellent substrate for sulphur incorporation since they contain a high number of functionalities and possess linear carbon skeletons (Moers et al., 1988). The compounds formed may yield thiophenes as stable thermal products upon pyrolysis. The ubiquitous occurrence of C5-C8 thiophenes in pyrolysates of sediments (e.g. Sinninghe Damsté et al., 1989) may thus be explained by sulphur incorporation into carbohydrates.
It is possible to incorporate sulphur into the macromolecular matrix of algal material of Phaeocystis sp.. Probably the sulphur has reacted with the algal carbohydrates resulting in the
formation of LMW OSC linked to the MM, which form C5-C8 thiophenes upon heating/pyrolysis.
Fukushima, K., Yasukawa, M., Muto, N., Uemera, H. & Ishiwatari, R., Org. Geochem. 18, 83-91 (1992).
Moers, M.E.C., de Leeuw, J.W., Cox, H.C. & Schenck, P.A., In Adv. Org. Geochem. 1987 (Novelli, L. & Matavelli, L., eds.) 1087-1091 (1988).
Rousseau, V., Mathot, S. & Lancelot, C., Mar. Biol. 107, 305-314 (1990).
Schouten , S., van Driel, G.B., Sinninghe Damsté, J.S. & de Leeuw, J.W., Geochim. et Cosmochim. Acta 57, 5111-5116 (1993).
Sinninghe Damsté, J.S., Eglinton, T.I.., de Leeuw, J.W. & Schenck, P.A., Geochim. et Cosmochim. Acta 53, 873-889 (1989).
Fig. 1: Partial total ion current of flash pyrolysis GC-MS; major peaks indicated; ![]() = thiophenes, number of carbon atoms indicated (C5-C8); a) residue of Phaeocystis sp. (after extraction etc.); b) residue of hydrosulphurized Phaeocystis sp. (after extraction etc.).
= thiophenes, number of carbon atoms indicated (C5-C8); a) residue of Phaeocystis sp. (after extraction etc.); b) residue of hydrosulphurized Phaeocystis sp. (after extraction etc.).